- Water Testing Meters
- Anemometer
- Length & Distance Meter
- Multimeter & Clamp Meter
- Light and Sound Meter
- Slide Calipers & Screw Gauge
- Thermometer & Hygrometer
- Milk Testing Meters
- Paper, Grain & Wood Testers
- Stopwatch & Timers
- Soil Testing Meters
- Refractometers & Analyzer
- Magnetic Compass
- Tachometer & Megger
- Thickness & Dia-Meters
- Other Meter And Accessories
Applications of Hydrochloric Acid (HCl) in Bangladesh

Hydrochloric Acid is a foamable Acid that is produced from Hydrogen Chloride Gas. It is also called HCl acid and muriatic acid in Bangladesh. There are some different grades and different hydrochloric acid price in Bangladesh.
When the gas hydrogen chloride is dissolved in water a strong chemical compound is produced called Hydrochloric Acid or HCl Acid.
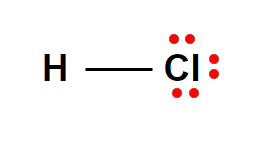
HCl is a colorless and odorless chemical solution of Hydrogen, chloride, and water. HCl acid is also known as muriatic acid and spirit of salt or acid salts. HCl acid is a highly corrosive chemical compound and it has lots of uses and applications in Bangladesh.
Key Applications of Hydrochloric Acid in Bangladesh
Metalworking, Food, and Nutrition, Laboratory chemicals, Leather processing, Water treatment, Cleaning products, Metal Finishing, Oil and Gas, pH control, etc.
Let’s see some major applications of hydrochloric acid in the broad sense
(A) Apply for the production of organic compounds
Hydrochloric acid or HCl acid has many industrial applications. One of the applications is the production of dichloroethane and vinyl chloride for organic compounds such as PVC. HCl is also used to make other organic compounds such as bisphenol A, which is used in various pharmaceutical products.
(B) For the production of inorganic compounds
Hydrochloric acid or HCl acid is highly used in the preparation of inorganic compounds. Water treatment chemicals such as poly aluminum chloride (PAC) and iron (III) chloride are used as coagulation and flocculation agents in drinking water production, sewage treatment, and paper production.
(C) Used to Remove the Metal Stains
Hydrochloric acid is a very corrosive acid. It is used to remove stains from metal. It can clean iron, copper, brass, and other metals; however, one should dilute it by adding 9 parts of water to 1 part of acid. It should not be used directly on metal as it is a very strong cleaning agent.
(D) Hydrochloric Acid for Cleaning Pools
Although pools can be cleaned with simple detergents and scrubs, some stains that stick to tiles are not easy to remove. In that case, hydrochloric acid comes in handy. Here too, 1 part acid to 10 parts water should be added to make a pool-cleaning solution.
(E) Used for Digestion of Food
Gastric juice in the human stomach also contains hydrochloric acid which helps in the digestion of food. It acidifies the contents of the stomach.
(F) Apply for Refining table salt
Hydrochloric acid or HCl acid is used to purify table salt in Pharmaceutical Industries.
(G) For neutralization and pH control
It is used to control the acidity (pH) of solutions. It is used to control the pH of pharmaceutical products, food, and drinking water. HCl is also used to neutralize waste streams (containing alkaline substances).
(H) Used for Regeneration of Ion Exchangers
HCl is used in the regeneration of ion exchange resins. It is used to leach cations from resins. Demineralized water and ion exchangers are used in drinking water production, all chemical industries, and many food industries in Bangladesh.
(I) Application of Hydrochloric Acid in oil Production
Hydrochloric acid is used in the oil production process. When the acid is injected into the rock, it creates a large-pore structure, which stimulates oil production.
(J) Used in Laboratory for Research and Analysis
Most schools, colleges, and universities use Hydrochloric Acid for Different types of Practical Experiments and for cleaning lab equipment.
Recommendation:
If you have any requirements for Hydrochloric Acid (HCl), you can contact to this email Labtexbangla@gmail.com with the Proper Institutional Documents and Written Purpose.
The above information was Sourced from byjus.com, iStock, studiousguy.com, etc.
