- Water Testing Meters
- Anemometer
- Length & Distance Meter
- Multimeter & Clamp Meter
- Light and Sound Meter
- Slide Calipers & Screw Gauge
- Thermometer & Hygrometer
- Milk Testing Meters
- Paper, Grain & Wood Testers
- Stopwatch & Timers
- Soil Testing Meters
- Refractometers & Analyzer
- Magnetic Compass
- Tachometer & Megger
- Thickness & Dia-Meters
- Other Meter And Accessories
Introduction to Periodic Table and its Significance
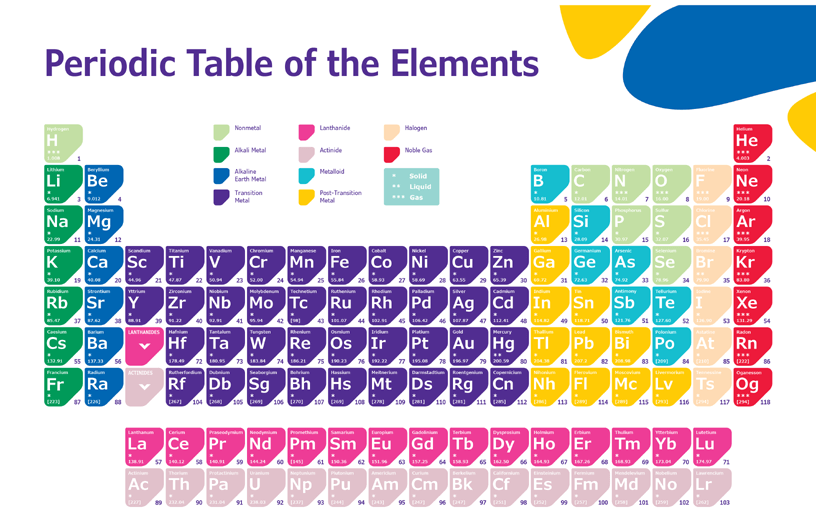
A periodic table is also called the periodic table of elements. A row and column arrangement of chemical elements in a word. It is mostly used in chemistry, physics, and other sciences. It is seen as an icon of chemistry.
What are the elements of the periodic table?
The simple or basic form of a pure substance that cannot be further broken down by any physical or chemical process. Also, elements cannot be created or synthesized from simple substances. That is, the basic form of a pure substance is the element of the periodic table. (Click to See 3D Periodic Table)
- A piece of gold can be broken into the smallest possible pieces, but the original element of gold cannot be further broken down or synthesized from some other substance. So gold is a periodic table element and so are iron, copper, silver, etc.
- H2O (water) is not an element, as it can be split into H (Hydrogen) and O (Oxygen). But oxygen and hydrogen can no longer be broken down so they are elements; So we can say that all compounds are made up of elements.
- Elements are the building blocks of all matter – solids, liquids, and gases. That is, all things in the universe are made up of basic elements.
“Amendment” The above-mentioned definition is no longer completely correct because after the discovery of radioactivity, it is known that elements can be broken down further, and also many elements have been synthesized in the laboratory from simpler substances.
Now elements are defined as pure substances that are made up of only one type of atom.
What is the periodic table?
As we know, there are 26 English alphabets and millions of books have been written using this alphabet. Alphabets combine to form different words that express different meanings. Similarly, scientists have discovered 118 elements that make up every known thing in the universe. These elements combine in different ways to form different substances. Just as the 26 English alphabets have a specific order, these 118 elements are arranged in a specific style based on their physical and chemical properties, this arrangement is called the periodic table. This arrangement is arranged according to the atomic number i.e. the number of protons in the element.

Some Interesting Things about Periodic Table Elements
Out of 118 elements 90 are found in nature and the remaining 28 are synthesized in Laboratory.
Most of the Elements Exist in Solid State at Room Temperature
Mercury and bromine are the only two elements that exist in the liquid state at room temperature
11 elements of the periodic table exist in the gaseous state at room temperature. Hydrogen, Nitrogen, Oxygen, Fluorine, Chlorine, Helium, Neon, Argon, Krypton, Xenon, and Radon.
More numbers of elements are metals than non-metals and metalloids.
Gallium and Cesium have very low melting points and exist in liquid storage just above normal room temperature.
Oxygen is the most abundant element in the earth’s crust followed by silicon and aluminum.
Nitrogen is the most abundant element in the atmosphere.
Hydrogen is the most abundant element in the universe followed by Helium and oxygen.
